Greenhouse effect Lab
Collaborators-
Griffin Matthews and Naara Mendez
Griffin Matthews and Naara Mendez
Abstract-
We conducted an experiment to find out what environmental conditions effect the greenhouse effect that takes place on Earth. We placed 2 thermometers, one inside and one outside of a an overturned beaker which was placed under a heat lamp. Our hypothesis was that both thermometers would heat up evenly and when the heat lamp was turned off and removed, the space inside the beaker would hold the heat longer than outside. We thought that because the air inside the beaker was enclosed, the air would remain heated, much like the greenhouse effect on earth. We did not get our desired results. The overturned beaker seemed to make little to no noticeable in the air temperature at all as both thermometers increased and decreased in temperature at the same rate.
We conducted an experiment to find out what environmental conditions effect the greenhouse effect that takes place on Earth. We placed 2 thermometers, one inside and one outside of a an overturned beaker which was placed under a heat lamp. Our hypothesis was that both thermometers would heat up evenly and when the heat lamp was turned off and removed, the space inside the beaker would hold the heat longer than outside. We thought that because the air inside the beaker was enclosed, the air would remain heated, much like the greenhouse effect on earth. We did not get our desired results. The overturned beaker seemed to make little to no noticeable in the air temperature at all as both thermometers increased and decreased in temperature at the same rate.
Problem-
What conditions cause the warming of the atmosphere?
What conditions cause the warming of the atmosphere?
Hypothesis-
If we place a heat lamp over an overturned beaker so the lamp heats both the air in the beaker and the air outside the beaker, then the temperature of the air inside the beaker will hold the heat longer than the air on the outside when we turn off and remove the heat lamp.
If we place a heat lamp over an overturned beaker so the lamp heats both the air in the beaker and the air outside the beaker, then the temperature of the air inside the beaker will hold the heat longer than the air on the outside when we turn off and remove the heat lamp.
Parts of the Experiment-
Control- The thermometer located outside the beaker. This is the unchanged temperature.
Independent Variable- The beaker. The glass should hold the heat longer, therefore manipulating the temperature.
Dependent Variable- The temperature inside the beaker. This temperature is dependent on the beaker being there.
Control- The thermometer located outside the beaker. This is the unchanged temperature.
Independent Variable- The beaker. The glass should hold the heat longer, therefore manipulating the temperature.
Dependent Variable- The temperature inside the beaker. This temperature is dependent on the beaker being there.
Materials-
Sheet of White Paper (2)
Tape
Beaker
Heat Lamp
Thermometer (2)
Heat Lamp Stand
Paper Towel (1/8 sheet)
Sheet of White Paper (2)
Tape
Beaker
Heat Lamp
Thermometer (2)
Heat Lamp Stand
Paper Towel (1/8 sheet)
Methods-
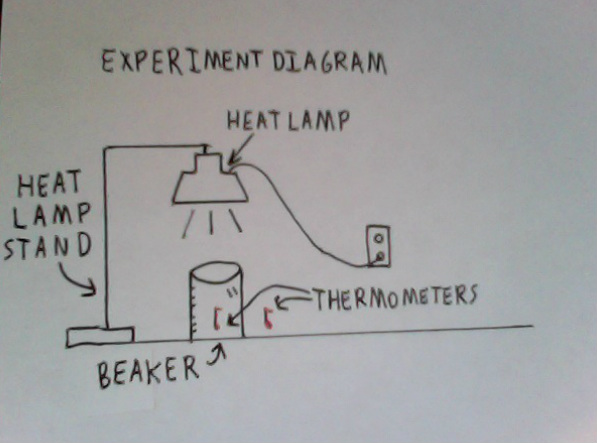
1. Start by putting white copy paper down on the table surface, taping the edges down to prevent slipping.
2. Place the overturned beaker on the paper with a thermometer inside. Also, place a thermometer a few inches outside the beaker.
2a. (If needed) Our beaker had a spout around the lip which was meant for liquids to be poured. This would allow air to flow freely in and out of the beaker. We remedied this by placing a folded paper towel in the spout when the beaker was turned over.
3. Set up the heat lamp stand and center the heat lamp over the overturned beaker and outside thermometer so the heat lamp shines light and heat evenly over both thermometers.
4. Turn on the heat lamp and leave it on until the thermometers have increased by at least 10 degrees, in our case 18 minutes to climb 12 degrees. Record temperature readings throughout the heating.
5. After reaching the desired temperature increase, remove the heat lamp to stop the lamp from continuing to radiate heat which will throw off the results.
6. Observe the data, recording temperature readings of both thermometers as they decrease.
1. Start by putting white copy paper down on the table surface, taping the edges down to prevent slipping.
2. Place the overturned beaker on the paper with a thermometer inside. Also, place a thermometer a few inches outside the beaker.
2a. (If needed) Our beaker had a spout around the lip which was meant for liquids to be poured. This would allow air to flow freely in and out of the beaker. We remedied this by placing a folded paper towel in the spout when the beaker was turned over.
3. Set up the heat lamp stand and center the heat lamp over the overturned beaker and outside thermometer so the heat lamp shines light and heat evenly over both thermometers.
4. Turn on the heat lamp and leave it on until the thermometers have increased by at least 10 degrees, in our case 18 minutes to climb 12 degrees. Record temperature readings throughout the heating.
5. After reaching the desired temperature increase, remove the heat lamp to stop the lamp from continuing to radiate heat which will throw off the results.
6. Observe the data, recording temperature readings of both thermometers as they decrease.
Data-
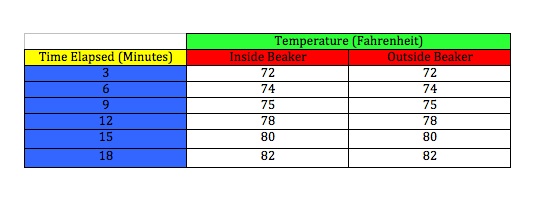
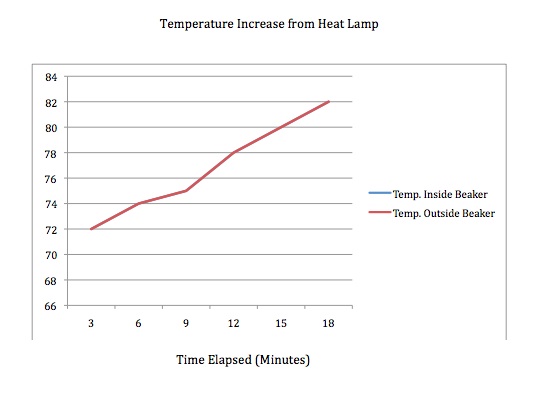
Heating up both thermometers
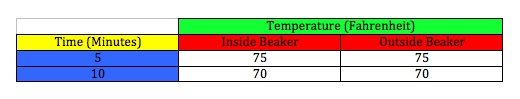
Cooling down both thermometers after heat lamp has been turned off and removed
Data Analysis-
Our data was somewhat unexpected. As you can see in the charts and graphs, the beaker made no difference in the heat retention of the air inside. The only difference not shown in the cooling chart is that towards the end of the temperature decrease, at about 10 minutes after the lamp was removed, we did notice a slight temperature difference between the two. It was too insignificant to note in the chart above and was a half a degree at most, but I felt it was worth noting here. These results reflect that the glass beaker is not a suitable substitution for the Earth's atmosphere. I think the key to making this experiment work is to find a good material that acts like the Earth's outer atmosphere. This experiment should work as the Greenhouse Effect is an almost certain theory.
Our data was somewhat unexpected. As you can see in the charts and graphs, the beaker made no difference in the heat retention of the air inside. The only difference not shown in the cooling chart is that towards the end of the temperature decrease, at about 10 minutes after the lamp was removed, we did notice a slight temperature difference between the two. It was too insignificant to note in the chart above and was a half a degree at most, but I felt it was worth noting here. These results reflect that the glass beaker is not a suitable substitution for the Earth's atmosphere. I think the key to making this experiment work is to find a good material that acts like the Earth's outer atmosphere. This experiment should work as the Greenhouse Effect is an almost certain theory.
Conclusion-
I'll begin with a quick review of what the Greenhouse Effect is. The Greenhouse Effect is when the Earth's atmosphere stores heat and regulates the planet's temperature. Our experiment tried to replicate this, however, we did not get our desired results. I found a blog post on Scientific American by David Wogan (Link to Article) which discusses the experiment Joseph Fourier used to prove his discovery of the Greenhouse Effect. He used a box with a glass cover heated by a lamp and his experiment worked with the air in the box retaining the heat. Our experiment was remarkably similar to his, yet did not yield the same results. I think a reason our experiment might not have worked could have been the fact that our overturned beaker was not air tight. The pouring spout definitely didn't help although we did the best we could at the time with the paper towel. Other groups used plastic wrap to seal the beaker which, looking back, would have been a better alternative than simply flipping the beaker over. Also discussed in the article, I think it is worth noting that Fourier's experiment was "overly simple" and that the physics of the glass holding the heat in is quite a bit different than the physics of Carbon Dioxide and Oxygen retaining heat. With the allotted time and resources available I believe our experiment could have been a great small-scale representation of the Greenhouse Effect. I think a second, more improved trial would yield in better results.
I'll begin with a quick review of what the Greenhouse Effect is. The Greenhouse Effect is when the Earth's atmosphere stores heat and regulates the planet's temperature. Our experiment tried to replicate this, however, we did not get our desired results. I found a blog post on Scientific American by David Wogan (Link to Article) which discusses the experiment Joseph Fourier used to prove his discovery of the Greenhouse Effect. He used a box with a glass cover heated by a lamp and his experiment worked with the air in the box retaining the heat. Our experiment was remarkably similar to his, yet did not yield the same results. I think a reason our experiment might not have worked could have been the fact that our overturned beaker was not air tight. The pouring spout definitely didn't help although we did the best we could at the time with the paper towel. Other groups used plastic wrap to seal the beaker which, looking back, would have been a better alternative than simply flipping the beaker over. Also discussed in the article, I think it is worth noting that Fourier's experiment was "overly simple" and that the physics of the glass holding the heat in is quite a bit different than the physics of Carbon Dioxide and Oxygen retaining heat. With the allotted time and resources available I believe our experiment could have been a great small-scale representation of the Greenhouse Effect. I think a second, more improved trial would yield in better results.
Citations-
http://blogs.scientificamerican.com/plugged-in/2013/05/16/why-we-know-about-the-greenhouse-gas-effect/ - David Wogan's article on Joseph Fourier's experiment, referenced in the conclusion above.
http://blogs.scientificamerican.com/plugged-in/2013/05/16/why-we-know-about-the-greenhouse-gas-effect/ - David Wogan's article on Joseph Fourier's experiment, referenced in the conclusion above.